
Estimating the effective reproduction number in real-time for a single timeseries with reporting delays
Epinowcast Team
Source:vignettes/single-timeseries-rt-estimation.Rmd
single-timeseries-rt-estimation.RmdThis vignette is partly based on this nowcasting example by Sebastian Funk and Sam Abbott.
In this case study we walk through a simple approach to jointly estimating the effective reproduction number over time and the delay from a positive test to this test being reported. For more on the epinowcast package see the documentation.
Use case
An epidemic is in progress. We want to know what the effective reproduction number is at the current time. This is the average number of secondary infections caused by a single infected individual at a given point in time and is a measure of the transmissibility of the infection in the population at that time.
Unfortunately, we can’t directly observe infections and the data we do observe suffers from reporting delays.
What we have
- A linelist of cases with a date of interest (for example test positive date) and a date of report (for example date of hospitalisation report);
- An estimate of the distribution of times from infection to positive test;
- An estimate of the generation time distribution, which is the distribution of the time intervals between the infection of a primary case and the infection of secondary cases caused by the primary case.
- An estimate of the ascertainment rate, which is the proportion of infections that are reported as hospitalisations.
What do we do
- Visualise and contextualise the data this case study uses (COVID-19 hospitalisations in Germany);
- Specify a joint model of the effective reproduction number, the delay from infection to hospitalisation and the delay from hospitalisation to report;
- Fit this model to the data available in real-time, retrospectively with delay estimation, and retrospectively without delay estimation;
- Visualise the nowcast from the real-time data in comparison to the data that was ultimately observed;
- Compare real-time, retrospective, and retrospective (no delay) reproduction number estimates and delay distribution estimates;
- Outline the limitations and strengths of this approach and highlight areas where it can be extended.
epinowcast is still in development and so the following case study has pain points and limitations that we are working on improving. If you have any suggestions or feedback please get in touch via the issues page. We also welcome contributions via pull requests (see our contributing guidelines.
Getting setup
See the package documentation for guidance on installing the package and getting setup with cmdstanr (the backend used here for fitting models).
As well as the epinowcast package we will use the following packages in this vignette.
Code
- Here we make use of
data.tablefor simple data transformations but tools from the tidyverse or base R tools could just as easily be used; - We also use
purrrto make use of thepartial()function which allows us to partially specify a function for later reuse. This is useful for specifying the model as we can specify the parts of the model that are common to all models and then specify the parts that are specific to each model when we call the updated function; - We use
ggplot2for plotting. - We use
primarycensoredfor simulating the primary censored and right truncated processes need to correctly model the discrete delays.
Introducing the data: COVID-19 hospitalisations in Germany
Overview
In this case study, we use data sourced from the Robert Koch Institute via the Germany Nowcasting hub. The data represent hospitalisation counts by date of positive test and date of test report in Germany. These data have been used extensively for nowcasting and forecasting COVID-19 hospitalisations in Germany and are described in detail in Wolffram et al. (2023). We use these data as they are a good example of the type of data that are available in many settings. We first summarise the data,
Code
summary(germany_covid19_hosp)
#> reference_date location age_group confirm
#> Min. :2021-04-06 DE : 90405 00-04:219555 Min. : 0.00
#> 1st Qu.:2021-05-15 DE-BB : 90405 00+ :219555 1st Qu.: 0.00
#> Median :2021-06-23 DE-BE : 90405 05-14:219555 Median : 1.00
#> Mean :2021-06-25 DE-BW : 90405 15-34:219555 Mean : 11.44
#> 3rd Qu.:2021-08-02 DE-BY : 90405 35-59:219555 3rd Qu.: 6.00
#> Max. :2021-10-20 DE-HB : 90405 60-79:219555 Max. :1552.00
#> (Other):994455 80+ :219555
#> report_date
#> Min. :2021-04-06
#> 1st Qu.:2021-06-24
#> Median :2021-08-03
#> Mean :2021-07-31
#> 3rd Qu.:2021-09-11
#> Max. :2021-10-20
#> In this case study we only consider national level data without stratification by age group. We can filter these data using data.table as follows,
Code
germany_hosp <- germany_covid19_hosp[location == "DE"][age_group == "00+"]
germany_hosp <- germany_hosp[, .(reference_date, report_date, confirm)]
germany_hosp
#> reference_date report_date confirm
#> <IDat> <Date> <int>
#> 1: 2021-04-06 2021-04-06 149
#> 2: 2021-04-07 2021-04-07 312
#> 3: 2021-04-08 2021-04-08 424
#> 4: 2021-04-09 2021-04-09 288
#> 5: 2021-04-10 2021-04-10 273
#> ---
#> 12911: 2021-07-27 2021-10-16 81
#> 12912: 2021-07-28 2021-10-17 159
#> 12913: 2021-07-29 2021-10-18 145
#> 12914: 2021-07-30 2021-10-19 117
#> 12915: 2021-07-31 2021-10-20 132Data transformations
These data are already in a format that can be used with epinowcast, as it contains
- a reference date (column
reference_date): the date of the observation, in this example the date of a positive test - a report date (column
report_date): the date of report for a given set of observations by reference date - a count (column
confirm): the total number of hospitalisations by reference date and report date. Note that this is cumulative by report date.
The package also provides a range of tools to convert data from line list, incidence, or other common formats into the required format (see Data converters). For example we could convert the data to a individual case linelist,
Code
germany_covid19_hosp_linelist <- germany_covid19_hosp |>
enw_filter_reference_dates_by_report_start() |>
enw_add_incidence() |>
enw_incidence_to_linelist()
germany_covid19_hosp_linelist
#> id reference_date location age_group report_date delay
#> <int> <IDat> <fctr> <fctr> <IDat> <int>
#> 1: 1 2021-04-06 DE 00+ 2021-04-06 0
#> 2: 2 2021-04-06 DE 00+ 2021-04-06 0
#> 3: 3 2021-04-06 DE 00+ 2021-04-06 0
#> 4: 4 2021-04-06 DE 00+ 2021-04-06 0
#> 5: 5 2021-04-06 DE 00+ 2021-04-06 0
#> ---
#> 11486202: 11486202 2021-10-20 DE-TH 15-34 2021-10-20 115
#> 11486203: 11486203 2021-10-20 DE-TH 60-79 2021-10-20 117
#> 11486204: 11486204 2021-10-20 DE-TH 60-79 2021-10-20 117
#> 11486205: 11486205 2021-10-20 DE-TH 60-79 2021-10-20 117
#> 11486206: 11486206 2021-10-20 DE-TH 60-79 2021-10-20 117This linelist could then be itself converted into the format epinowcast requires using the enw_linelist_to_incidence() function,
Code
incidence_from_linelist <- enw_linelist_to_incidence(
germany_covid19_hosp_linelist,
reference_date = "reference_date",
report_date = "report_date",
by = c("age_group", "location"),
max_delay = 30
)
incidence_from_linelist
#> Key: <age_group, location, reference_date, report_date>
#> age_group location report_date reference_date new_confirm confirm
#> <fctr> <fctr> <IDat> <IDat> <int> <int>
#> 1: 00-04 DE-BY 2021-04-06 <NA> 0 0
#> 2: 00-04 DE-BY 2021-04-07 <NA> 0 0
#> 3: 00-04 DE-BY 2021-04-08 <NA> 0 0
#> 4: 00-04 DE-BY 2021-04-09 <NA> 0 0
#> 5: 00-04 DE-BY 2021-04-10 <NA> 0 0
#> ---
#> 2049593: 80+ DE-TH 2021-10-19 2021-10-18 0 0
#> 2049594: 80+ DE-TH 2021-10-20 2021-10-18 0 0
#> 2049595: 80+ DE-TH 2021-10-19 2021-10-19 1 1
#> 2049596: 80+ DE-TH 2021-10-20 2021-10-19 0 1
#> 2049597: 80+ DE-TH 2021-10-20 2021-10-20 0 0
#> delay
#> <int>
#> 1: 0
#> 2: 1
#> 3: 2
#> 4: 3
#> 5: 4
#> ---
#> 2049593: 1
#> 2049594: 2
#> 2049595: 0
#> 2049596: 1
#> 2049597: 0Here we have specified the reference_date and report_date columns, the by columns (which are used to group the data), and the max_delay which is the maximum delay between the reference date and the report date (note that the observed delay is longer than our specified delay and so is used instead). The enw_linelist_to_incidence() function will then calculate the incidence by reference date and report date for each group.
Filtering the data
For this case study we will only consider data from the 1st of March 2020 onwards. As a first step, we filter the data to only include observations from the 1st of May 2021 until the 1st of August 2021. We do this using the enw_filter_report_dates() and enw_filter_reference_dates() functions,
Code
complete_germany_hosp <- germany_hosp |>
enw_filter_report_dates(latest_date = "2021-08-01") |>
enw_filter_reference_dates(earliest_date = "2021-05-01") |>
enw_complete_dates(missing_reference = FALSE) |>
enw_filter_reference_dates_by_report_start() |>
enw_add_incidence()Here we have also used enw_complete_dates() to make sure that we have complete data for all dates between the earliest and latest dates. We have also used enw_add_incidence() to add the incidence column to the data.
Next we split the data into two parts, the data that was available at the time and that will be used to fit the model (rt_nat_germany) and the data that was available retrospectively about the same time period will be used to evaluate the model (retro_nat_germany). We again do this using the enw_filter_report_dates() and enw_filter_reference_dates() functions as follows:
- Create the real-time dataset (
rt_nat_germany) by filtering the data to only include reported observations from the 1st of May 2021 until the 1st of July 2021;
Code
rt_germany <- complete_germany_hosp |>
enw_filter_report_dates(latest_date = "2021-07-01")
rt_germany
#> report_date reference_date confirm new_confirm delay
#> <IDat> <IDat> <int> <int> <int>
#> 1: 2021-05-01 2021-05-01 234 234 0
#> 2: 2021-05-02 2021-05-01 352 118 1
#> 3: 2021-05-03 2021-05-01 391 39 2
#> 4: 2021-05-04 2021-05-01 464 73 3
#> 5: 2021-05-05 2021-05-01 507 43 4
#> ---
#> 1949: 2021-06-30 2021-06-29 31 11 1
#> 1950: 2021-07-01 2021-06-29 35 4 2
#> 1951: 2021-06-30 2021-06-30 20 20 0
#> 1952: 2021-07-01 2021-06-30 37 17 1
#> 1953: 2021-07-01 2021-07-01 20 20 0- Create the retrospective dataset (
retro_germany) by filtering the data to only include observations with reference dates (i.e. they may have been reported anytime up to the 1st of August 2021) from the 1st of May 2021 until the 1st of July 2021;
Code
retro_germany <- complete_germany_hosp |>
enw_filter_reference_dates(latest_date = "2021-07-01")
retro_germany
#> report_date reference_date confirm new_confirm delay
#> <IDat> <IDat> <int> <int> <int>
#> 1: 2021-05-01 2021-05-01 234 234 0
#> 2: 2021-05-02 2021-05-01 352 118 1
#> 3: 2021-05-03 2021-05-01 391 39 2
#> 4: 2021-05-04 2021-05-01 464 73 3
#> 5: 2021-05-05 2021-05-01 507 43 4
#> ---
#> 3871: 2021-07-28 2021-07-01 57 0 27
#> 3872: 2021-07-29 2021-07-01 57 0 28
#> 3873: 2021-07-30 2021-07-01 57 0 29
#> 3874: 2021-07-31 2021-07-01 57 0 30
#> 3875: 2021-08-01 2021-07-01 57 0 31Finally we can create a dataset that contains the latest data available for each reference date at the maximum modelled delay.
We do this using the enw_obs_at_delay() function, which combines enw_filter_delay() and enw_latest_data(),
Code
latest_germany_hosp <- retro_germany |>
enw_obs_at_delay(max_delay = 30)
head(latest_germany_hosp, n = 10)
#> reference_date .group report_date confirm new_confirm delay
#> <IDat> <num> <IDat> <int> <int> <int>
#> 1: 2021-05-01 1 2021-05-30 868 1 29
#> 2: 2021-05-02 1 2021-05-31 673 0 29
#> 3: 2021-05-03 1 2021-06-01 390 1 29
#> 4: 2021-05-04 1 2021-06-02 735 7 29
#> 5: 2021-05-05 1 2021-06-03 894 2 29
#> 6: 2021-05-06 1 2021-06-04 889 2 29
#> 7: 2021-05-07 1 2021-06-05 803 0 29
#> 8: 2021-05-08 1 2021-06-06 721 0 29
#> 9: 2021-05-09 1 2021-06-07 491 0 29
#> 10: 2021-05-10 1 2021-06-08 308 0 29Visualising the data
Before we define, or fit, a model we should visualise the data to get an idea of the trends in the data and its reporting structure. There is currently no function in epinowcast to visualise the data, but we can use the ggplot2 package to do this manually as follows,
Code
gh_vis_cohorts <- copy(retro_germany)[
,
report_date := fcase(
report_date <= as.Date("2021-05-15"), as.Date("2021-05-15"),
report_date <= as.Date("2021-06-01"), as.Date("2021-06-01"),
report_date <= as.Date("2021-06-15"), as.Date("2021-06-15"),
report_date <= as.Date("2021-07-01"), as.Date("2021-07-01"),
report_date <= as.Date("2021-07-15"), as.Date("2021-07-15"),
report_date <= as.Date("2021-08-01"), as.Date("2021-08-01")
) |>
factor(levels = rev(c(
"2021-05-15", "2021-06-01", "2021-06-15", "2021-07-01",
"2021-07-15", "2021-08-01"
)))
]
gh_vis_cohorts_by_reference <- gh_vis_cohorts[,
.(confirm = sum(new_confirm)),
by = .(reference_date)
]
gh_vis_cohorts_by_ref_rep <- gh_vis_cohorts[,
.(confirm = sum(new_confirm)),
by = .(reference_date, report_date)
]
gh_vis_cohorts_by_ref_rep |>
ggplot() +
aes(
x = reference_date, y = confirm, fill = report_date, group = report_date
) +
geom_col(position = "stack", alpha = 1, col = "grey") +
geom_vline(
aes(xintercept = as.Date(report_date)),
linetype = 2, alpha = 0.9
) +
xlim(c(
min(gh_vis_cohorts_by_ref_rep$reference_date),
max(gh_vis_cohorts_by_ref_rep$reference_date)
)) +
scale_y_continuous(labels = \(x)(scales::comma(x, accuracy = 1))) +
scale_fill_brewer(
palette = "Blues", aesthetics = c("color", "fill")
) +
theme_bw() +
labs(
x = "Date of positive test",
y = "Hospitalised cases by date of positive test",
fill = "Report date"
) +
guides(fill = guide_legend(reverse = TRUE)) +
theme(legend.position = "bottom")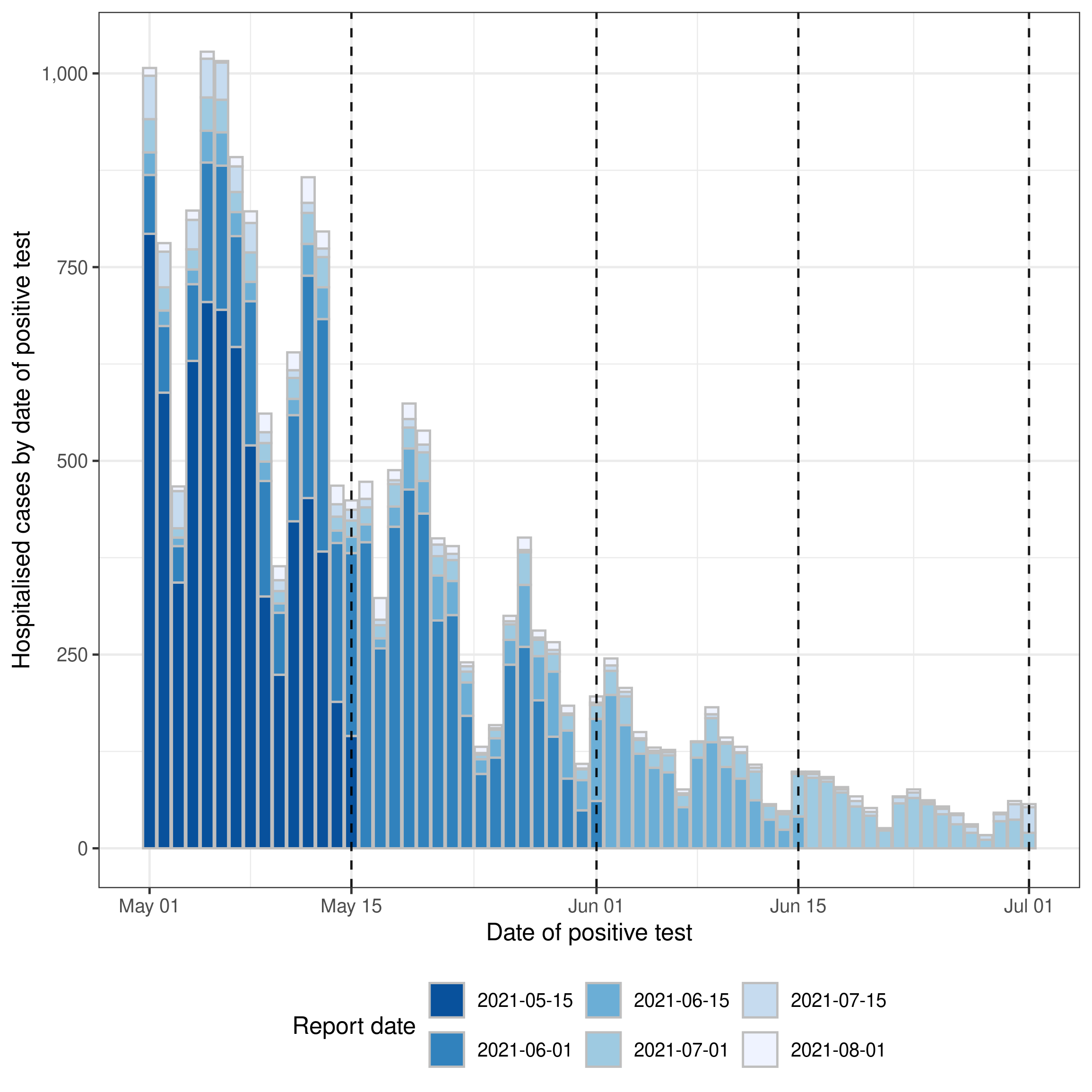
Model
The epinowcast package provides a flexible framework for modelling the incidence of infections and subsequent measures such as hospitalisations that are themselves reported with some delay. The framework is based on a generative model that describes the hypothesised process by which the data are generated. The generative model is then used to define a likelihood function that describes the probability of observing the data given the model parameters. The likelihood function is then combined with prior information used to fit the model parameters to the data. In the following sections we describe the generative model for the incidence of infections and hospitalisations. If you would like to know more about the general framework see the model vignette.
Expected hospitalisations
The first part of the generative process we consider is the expected number of hospitalisations by date positive test. This is the number of hospitalisations that we would expect to observe if we had perfect reporting of hospitalisations and there was no unmodelled variance. This part of the model is also decomposed into three parts: the expected number of infections, the delay from infection to positive test, and the probability of hospitalisation given an infection. These three parts are combined to give the expected number of hospitalisations by date positive test. We describe each of these parts in turn.
Expected infections
We model expected infections as a function of the effective (instantaneous) reproduction number and previous infections weighted by the generation time using the renewal equation[1,2]. This corresponds to the commonly used Susceptible-Exposed-Infected-Recovered (SEIR) model when the appropriate generation time is specified[3].
Instantaneous reproduction number
We model the instantaneous reproduction number (\(R_t\)) on the log scale as a weekly random walk as follows
\[\begin{align} \text{log} (R_{t}) &= r_0 + \sum_{i = 0}^{t \% 7} \epsilon_i \\ \epsilon_i &\sim \text{Normal}\left(0, \sigma_{\epsilon} \right) \\ \sigma_{\epsilon} &\sim \text{HalfNormal}\left(0, 1 \right) \end{align}\]
where \(r_0\) is the intercept, and \(\sum_{i = 0}^{t \% 7} \epsilon_i\) is a weekly random walk.
We choose this model because it is flexible, can be used to model a range of different scenarios, and is parsimonious. For example, if we set \(\sigma_{\epsilon} = 0\) then the model reduces to a constant reproduction number. If we set \(\sigma_{\epsilon} > 0\) then the model allows for changes in the reproduction number over time. Other sensible choices for the model include a random walk with a longer time scale (e.g. monthly) or shorter time scale (e.g. daily), a random walk with a drift term, or a random walk with a drift term and a seasonal component.
Latent infections
We model the expected number of infections/latent notifications (\(\lambda^l\)) using a renewal process[1,2]. This model is a generalisation of the default epinowcast model and implies that current infections/notifications are dependent on past infections/notifications based on a kernel. This kernel is defined by the generation time distribution, the probability distribution of the time between infection and subsequent infectiousness, and the previously defined model for the effective reproduction number. The model is defined as follows,
\[\begin{align} \lambda^l_{t} &\sim \text{LogNormal}\left(\mu^{l}, \sigma^{l} \right),\ t \leq P \\ \lambda^l_{t} &= R_{t} \sum_{p = 1}^{P} g\left(t - p \right) \lambda^l_{t-p},\ t \gt P \end{align}\]
We assume the generation time (\(g\)) is a gamma distribution with mean 4 days and a standard deviation of 3 days. To initialise the model we assume that the first \(P\) latent notifications are log-normally distributed with mean \(\mu^{l}_{g,t}\) and standard deviation \(\sigma^{l}_{g,t}\). This is equivalent to assuming that the first \(P\) latent notifications are independent and identically distributed. The mean of the log-normal distribution for each group is the log of the latest reported case count for the first reference date for that group scaled by the sum of the latent reporting delay. The standard deviation is assumed to be 1.
Latent reporting delay and ascertainment
In many settings, such as in this case study, there may be additional reporting delays on top of those that are directly observed in the data, and therefore “nowcastable”, a common example is the delay from exposure to symptom onset. For these settings we support modelling “latent” reporting delays as a convolution of the underlying expected counts with the potential for these delays to vary over time and by group. This implementation is similar to that implemented in EpiNow2 and epidemia as well as other similar models[4–7]. In addition to this, we support modelling ascertainment through the use of improper probability mass functions (i.e. by not enforcing a sum to 1 constraint) and inferring ascertainment where possible (for example day of the week reporting patterns).
Here we assume an infection to hospitalisation ratio of 2% modified by a random effect for the day of the week to account for within week reporting periodicity. We also assume a lognormal reporting delay with a mean of 5 days and a standard deviation of 2 days, representing the delay from infection to positive test. We assume that the reporting delay is the same for all days of the week. This model is defined as follows,
\[\begin{align} \lambda_{t} &= \nu_{t \% 7} \sum_{\tau = 0}^{L - 1} f_{g}\left(t - \tau \right) \lambda^l_{t - \tau} \\ \text{log} (\nu_{i}) &\sim \text{Normal}\left(-3.91, \sigma_{\nu} \right) \\ \sigma_{\nu} &\sim \text{HalfNormal}\left(0, 1 \right) \\ \end{align}\]
Where \(\nu_{i}\) is a random effect for each day of the week which represents reporting periodicity.
Specifying the model using epinowcast::enw_expectation()
We first need to define the weekly random walk model for the effective reproduction number. We can do this using the formula interface as follows,
Code
rt_formula <- ~ 1 + rw(week)Next we define the generation time distribution. As already discussed, we assume a gamma distribution with mean 4 days and a standard deviation of 3 days. These summary parameters first need to be transformed to the shape and scale parameters used for the gamma distribution. We then need to account for daily censoring and finally normalise the probability mass function (PMF) to account for right truncation.
Code
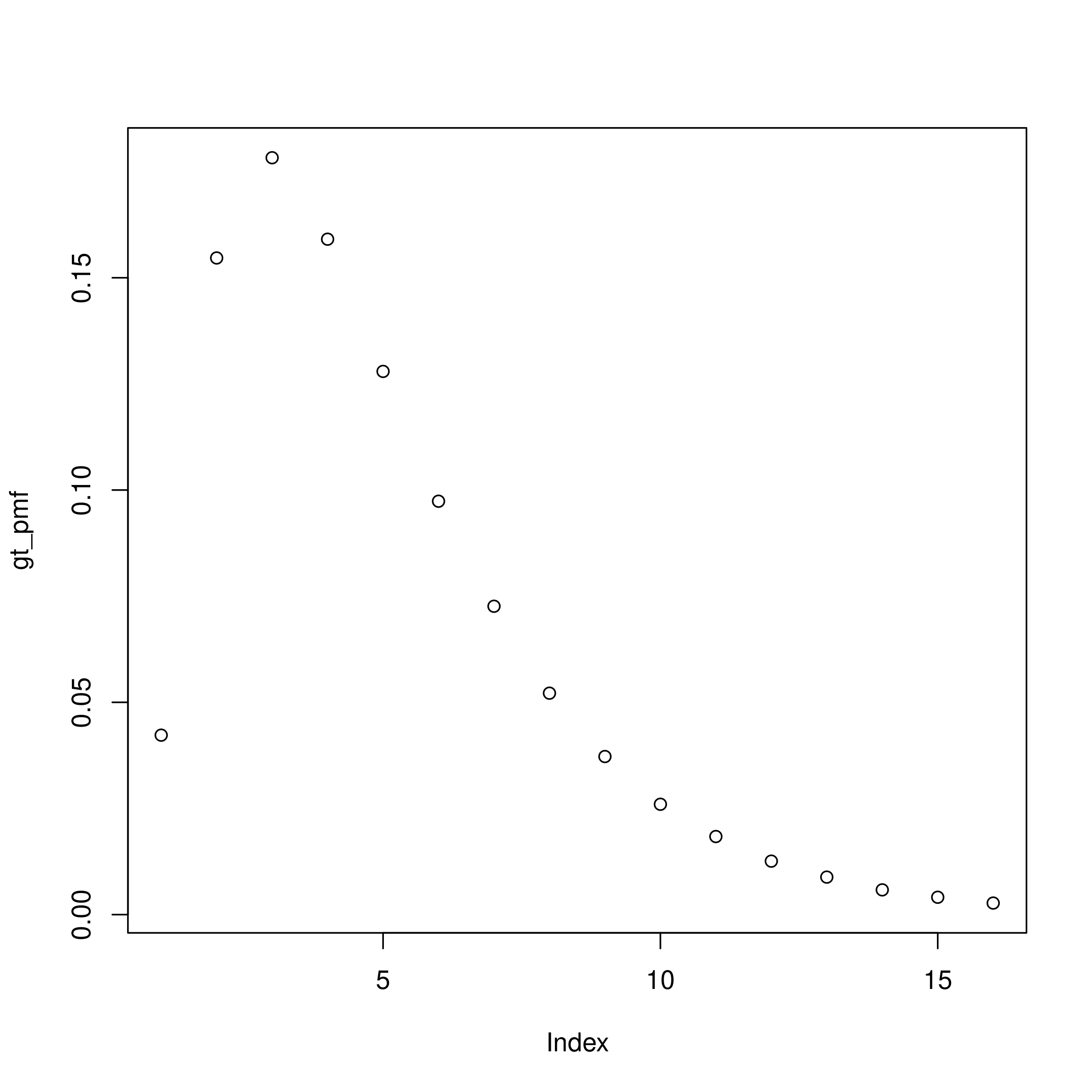
Here and for other PMFs we set the maximum delay to be 15 days as this gives good coverage of the majority of the distribution and limits the computational burden of fitting the model.
Now we define the latent reporting delay in a similar way remembering we specified a lognormal distribution with a mean of 5 days and a standard deviation of 2 days. We again also need to account for daily censoring and normalise the PMF to account for right truncation.
Code
lgn_mean <- 5
lgn_sd <- 2
meanlog <- log(lgn_mean^2 / sqrt(lgn_sd^2 + lgn_mean^2))
sdlog <- sqrt(log(1 + lgn_sd^2 / lgn_mean^2))
# then discretise the distribution
reporting_pmf <- 0:14 |>
dpcens(plnorm, meanlog = meanlog, sdlog = sdlog, D = 15) |>
# and scale it by the infection to hospitalisation ration (2%)
(\(x) x * 0.02)()
plot(reporting_pmf)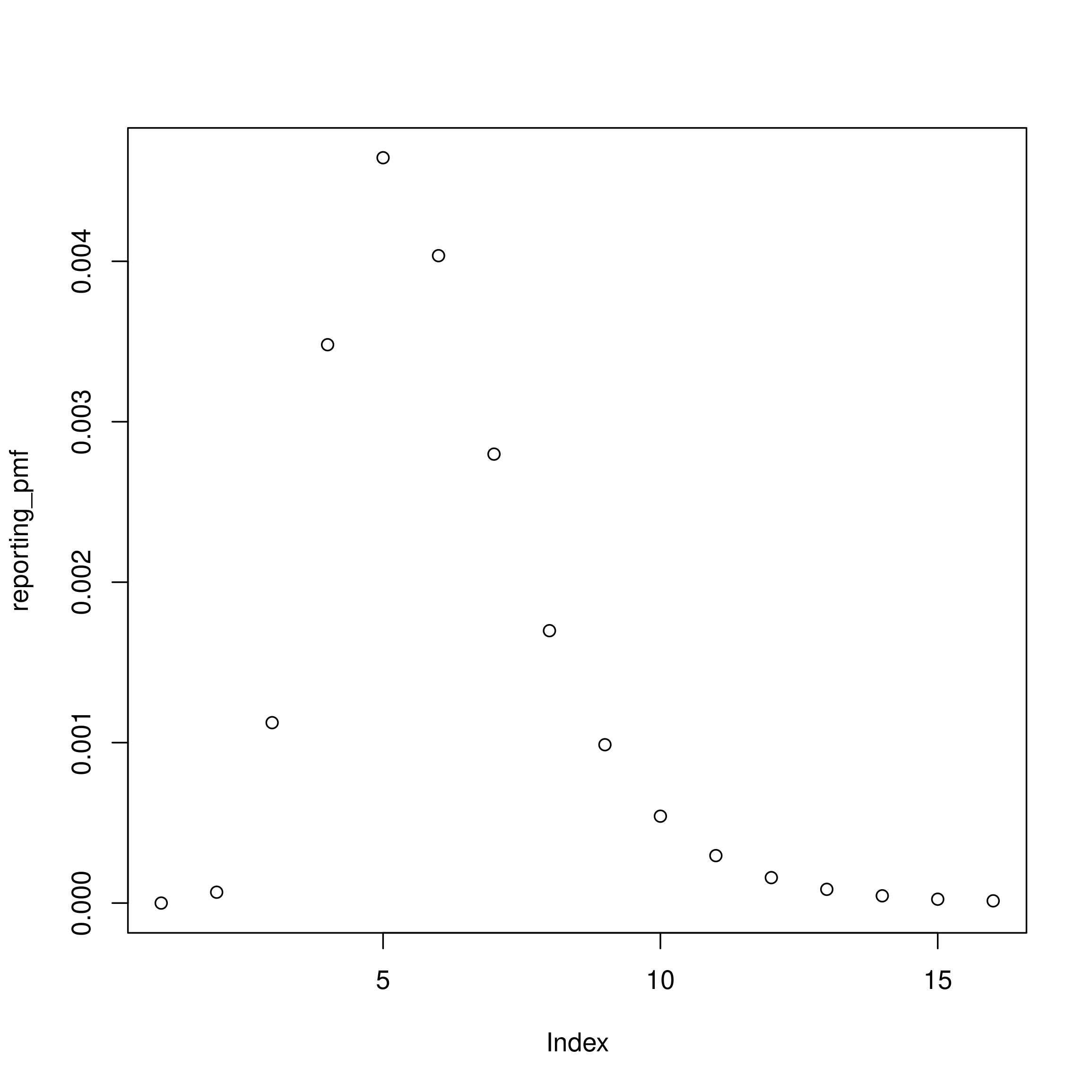
The last part of the model to define is the remaining part of the ascertainment model for expected hospitalisations. As we have already defined the base ascertainment ratio (i.e. the 2% infection to hospitalisation ratio) we only need to define the day of the week random effect. We can do this using the formula interface as follows,
Code
observation_formula <- ~ 1 + (1 | day_of_week)Finally, we can combine these four parts of the model to define the expected hospitalisations by date of positive test. We do this using the epinowcast::enw_expectation() function as follows,
Code
expectation_module <- partial(
epinowcast::enw_expectation,
r = rt_formula,
generation_time = gt_pmf,
latent_reporting_delay = reporting_pmf,
observation = observation_formula
)Delay distribution
Defining the delay distribution
Given case counts both by date of reference and by date of report, we can estimate the reporting delay distribution directly and jointly with the underlying process model, rather than relying on external estimates from other sources (though we may want to account for external information in our priors).
See ?epinowcast for details on inspecting and customising priors.
We consider the reporting delay to follow a \(\text{LogNormal} \left(\mu^d, \sigma^d \right)\) distribution, with parameters
\[\begin{align} \mu^d &\sim \text{Normal} \left(0, 1 \right) \\ \sigma^d &\sim \text{Half-Normal} \left(0, 1 \right). \end{align}\]
This distribution is discretised into daily probabilities \(p_{t,d}\) and adjusted for the maximum delay, see vignette("distributions") for details.
Specifying the model using epinowcast::enw_reference()
We can define the reporting delay distribution using the epinowcast::enw_reference() function as follows,
Code
reference_module <- partial(enw_reference, ~1, distribution = "lognormal")Observation model and nowcast
Defining the observation model
Expected notifications by date of positive test (\(t\)) and reporting date can now be found by multiplying expected final notifications by date of positive test for each \(t\) with the probability of reporting for each day of delay (\(p_{t,d}\)). We assume a Poisson observation model and produce a nowcast of final observed hospitalisations at each reference date by summing posterior estimates for unobserved notification and observed notifications for that reference date.
\[\begin{align} n_{t,d} \mid \lambda_{t},p_{t,d} &\sim \text{Poisson} \left(\lambda_{t} \times p_{t,d} \right),\ t=1,...,T. \\ N_{t} &= \sum_{d=0}^{D} n_{t,d} \end{align}\]
Specifying the model using epinowcast::enw_obs()
Code
obs_module <- partial(enw_obs, family = "poisson")Fitting the model to COVID-19 hospitalisations in Germany
Preprocess the data
Before fitting the model we have just defined we need to preprocess the data in order for it to be in the correct format to work with epinowcast and to produce metadata that describes aspects of the data we use in the model (for example the maximum delay, the number of groups, and the number of observations in each group). We do this using the enw_preprocess_data() function as follows for both the real-time and retrospective datasets. Note that we set max_delay = 30 to constrain the modelled maximum delay to 30 days. This is a pragmatic choice to ensure that the model can be fit in a reasonable amount of time, but we could also set this to be longer if we wanted to and if the data suggested a longer delay was possible.
Code
rt_germany_pobs <- enw_preprocess_data(rt_germany, max_delay = 30)
rt_germany_pobs
#> ── Preprocessed nowcast data ───────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 30
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-01
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 1,425 x 8
#> new_confirm : 1,425 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 32
#> metareference : 62 x 8
#> metareport : 91 x 10
#> metadelay : 30 x 5Individual datasets can be extracted with enw_get_data(), for example the latest observations:
Code
enw_get_data(rt_germany_pobs, "latest") |> head()
#> reference_date report_date .group max_confirm confirm delay
#> <IDat> <IDat> <num> <int> <int> <num>
#> 1: 2021-05-01 2021-05-30 1 941 868 29
#> 2: 2021-05-02 2021-05-31 1 724 673 29
#> 3: 2021-05-03 2021-06-01 1 413 390 29
#> 4: 2021-05-04 2021-06-02 1 773 735 29
#> 5: 2021-05-05 2021-06-03 1 969 894 29
#> 6: 2021-05-06 2021-06-04 1 966 889 29
#> cum_prop_reported prop_reported
#> <num> <num>
#> 1: 0.9224230 0.001062699
#> 2: 0.9295580 0.000000000
#> 3: 0.9443099 0.002421308
#> 4: 0.9508409 0.009055627
#> 5: 0.9226006 0.002063983
#> 6: 0.9202899 0.002070393We do the same for the retrospective data:
Code
retro_germany_pobs <- enw_preprocess_data(retro_germany, max_delay = 30)
retro_germany_pobs
#> ── Preprocessed nowcast data ───────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 30
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-30
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 1,860 x 8
#> new_confirm : 1,860 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 32
#> metareference : 62 x 8
#> metareport : 120 x 10
#> metadelay : 30 x 5We also create a retrospective dataset without reporting delays using enw_retrospective().
This converts the preprocessed data to max_delay = 1 by taking the latest observation for each reference date and treating it as the final count with no delay.
This is useful when we want to estimate Rt from data that is already (approximately) complete and do not need to model reporting delays.
Code
retro_nodelay_pobs <- enw_retrospective(retro_germany_pobs)
retro_nodelay_pobs
#> ── Preprocessed nowcast data ───────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 1
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-01
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 62 x 7
#> new_confirm : 62 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 3
#> metareference : 62 x 7
#> metareport : 62 x 10
#> metadelay : 1 x 5Fitting the epinowcast model
Specifying the fitting options
Before we are ready to fit the model we need to first specify some fitting options for using cmdstanr (more information on these options can be found in the cmdstanr documentation). We use 2 chains with 2 threads per chain (so using 4 CPU cores in total) 500 warmup samples and 1000 iterations per chain. As this is a complex model we set adapt_delta to 0.98 (the default is 0.8) so that the Hamiltonian Monte Carlo sampler can explore the posterior distribution more efficiently. For the same reason, we have also set the max_treedepth to 15 (the default is 10). Finally, we set save_warmup to FALSE to save space (we don’t need the warmup samples for this analysis), and pp to TRUE so that we can use posterior predictive checks to assess the model fit.
Code
fit_module <- partial(enw_fit_opts,
chains = 2,
parallel_chains = 2,
threads_per_chain = floor(min(max(1, parallel::detectCores() / 4), 2)),
iter_warmup = 500,
iter_sampling = 1000,
max_treedepth = 12,
save_warmup = FALSE,
pp = TRUE,
show_messages = interactive(), # set this to TRUE to show fitting messages
refresh = ifelse(interactive(), 100, 0) # remove this to show fitting progress
)If you have more than 4 CPU cores available you can increase the number of threads per chain to make use of these additional cores. For example if you have 8 CPU cores available you could set threads_per_chain = 4 to use all 8 cores (as there are 2 chains).
Compiling the model
We also need to compile the model using cmdstanr before fitting it (we only have to do this once after installing the package). In order for this line to work you should have followed the installation guide in the README. This will take around a minute to run when called for the first time.
Code
epinowcast_model <- enw_model()Fitting the model
Now we are ready to bring together all the modules we have specified, combine them with the model we have just compiled, and fit our synthetic data. We first fit to the data that would have been available in real-time,
Code
germany_nowcast <- epinowcast(
data = rt_germany_pobs,
expectation = expectation_module(data = rt_germany_pobs),
reference = reference_module(data = rt_germany_pobs),
obs = obs_module(data = rt_germany_pobs),
fit = fit_module(),
model = epinowcast_model
)
germany_nowcast
#> ── epinowcast model output ─────────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 30
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-01
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 1,425 x 8
#> new_confirm : 1,425 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 32
#> metareference : 62 x 8
#> metareport : 91 x 10
#> metadelay : 30 x 5
#>
#> Model objects (access with `enw_get_data(x, "<name>")`):
#> priors : 28 x 6
#> fit : CmdStanMCMC
#> data : list(115)
#> fit_args : list(9)
#> init_method_output : NULL
#> Model fit:
#> Samples: 2,000 | Max Rhat: 1.01
#> Divergent transitions: 0 (0%)
#> Max treedepth: 10 (12 at max, 0.6%)
#> Run time: 159.3 secsFitting this model with the default options will take around 5 minutes to run on a laptop with 4 CPU cores. If you have more CPU cores available you can reduce the time it takes to fit the model by increasing the number of threads per chain (see the fitting options section above).
and then on the retrospectively observed data.
Code
retro_germany_nowcast <- epinowcast(
data = retro_germany_pobs,
expectation = expectation_module(data = retro_germany_pobs),
reference = reference_module(data = retro_germany_pobs),
obs = obs_module(data = retro_germany_pobs),
fit = fit_module(),
model = epinowcast_model
)
retro_germany_nowcast
#> ── epinowcast model output ─────────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 30
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-30
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 1,860 x 8
#> new_confirm : 1,860 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 32
#> metareference : 62 x 8
#> metareport : 120 x 10
#> metadelay : 30 x 5
#>
#> Model objects (access with `enw_get_data(x, "<name>")`):
#> priors : 28 x 6
#> fit : CmdStanMCMC
#> data : list(115)
#> fit_args : list(9)
#> init_method_output : NULL
#> Model fit:
#> Samples: 2,000 | Max Rhat: 1.01
#> Divergent transitions: 0 (0%)
#> Max treedepth: 10 (27 at max, 1.4%)
#> Run time: 185.1 secsYou may see a number of warning messages from cmdstanr when fitting the model. These generally occur when the model is being initialised and are due to this process not currently being optimised in epinowcast. The model should still fit correctly unless these warnings continue into the fitting process (outside of the warmup phase). This is something we are actively working on improving in future versions of epinowcast.
Finally, we fit the model to the retrospective data without delay estimation.
Here we set parametric = ~0 and non_parametric = ~0 for both the reference and report date models, disabling the delay model entirely.
The model only estimates the expectation (Rt and latent process) from the final observation counts.
Code
retro_nodelay_nowcast <- epinowcast(
data = retro_nodelay_pobs,
expectation = expectation_module(data = retro_nodelay_pobs),
reference = enw_reference(
parametric = ~0, non_parametric = ~0,
data = retro_nodelay_pobs
),
report = enw_report(non_parametric = ~0, data = retro_nodelay_pobs),
obs = obs_module(data = retro_nodelay_pobs),
fit = fit_module(),
model = epinowcast_model
)
retro_nodelay_nowcast
#> ── epinowcast model output ─────────────────────────────────────────────────────
#> Groups: 1 | Timestep: day | Max delay: 1
#> Observations: 62 timepoints x 62 snapshots
#> Max date: 2021-07-01
#>
#> Datasets (access with `enw_get_data(x, "<name>")`):
#> obs : 62 x 7
#> new_confirm : 62 x 9
#> latest : 62 x 8
#> missing_reference : 0 x 4
#> reporting_triangle : 62 x 3
#> metareference : 62 x 7
#> metareport : 62 x 10
#> metadelay : 1 x 5
#>
#> Model objects (access with `enw_get_data(x, "<name>")`):
#> priors : 28 x 6
#> fit : CmdStanMCMC
#> data : list(115)
#> fit_args : list(9)
#> init_method_output : NULL
#> Model fit:
#> Samples: 2,000 | Max Rhat: 1.01
#> Divergent transitions: 0 (0%)
#> Max treedepth: 10 (51 at max, 2.5%)
#> Run time: 24.1 secsThis model fits much faster than the models with delay estimation as it has far fewer parameters and no delay convolution.
Visualising the Nowcast
Plotting the nowcast based on real-time data
We first plot the nowcast based on real-time data. Effectively here we are trying to use our model to estimate the hospitalisations that will ultimately be reported based on the data that is currently available. We can compare this to the most recent observations to see how well the model is doing.
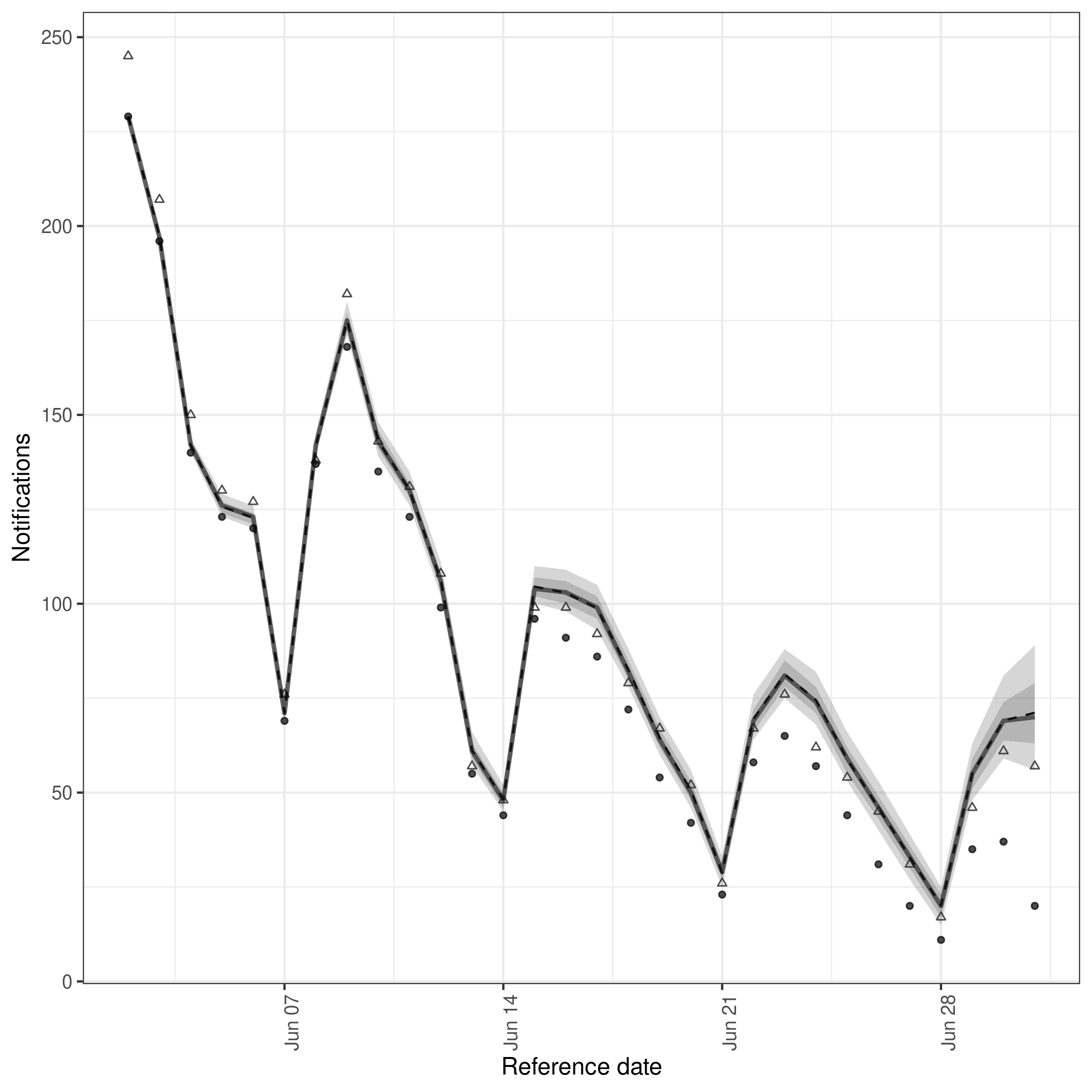
Internally, this plotting function (see ?plot.epinowcast()) sums observed data and posterior estimates for unobserved data to produce a nowcast of the number of hospitalisations that will ultimately be reported.
In this instance, the model has done a good job of estimating the number of hospitalisations that will ultimately be reported based on the data that is currently available. This is not always the case, and the model can sometimes over or under estimate the number of hospitalisations that will ultimately be reported.
An important thing to note here is that this model is clearly not perfect with more complete data being less well estimated by the model. This likely indicates some misspecification in the reporting delay model which could be fixed by making it more complex or even by making it entirely non-parametric (vs being based on a LogNormal).
Plotting the nowcast based on retrospective data
We can also plot the nowcast based on the retrospective data. As this plot uses observed data where available this is effectively plotting observed data after a 30 day delay (as this was the maximum we specified) compared to data that will ultimately be observed. This can be used to get a feel for how well specified our maximum delay is. Here we see that a small number of hospitalisations will be reported after 30 days. This will impact the performance of even an ideal model as it will not be able to predict these hospitalisations and so should be accounted for in the model evaluation.
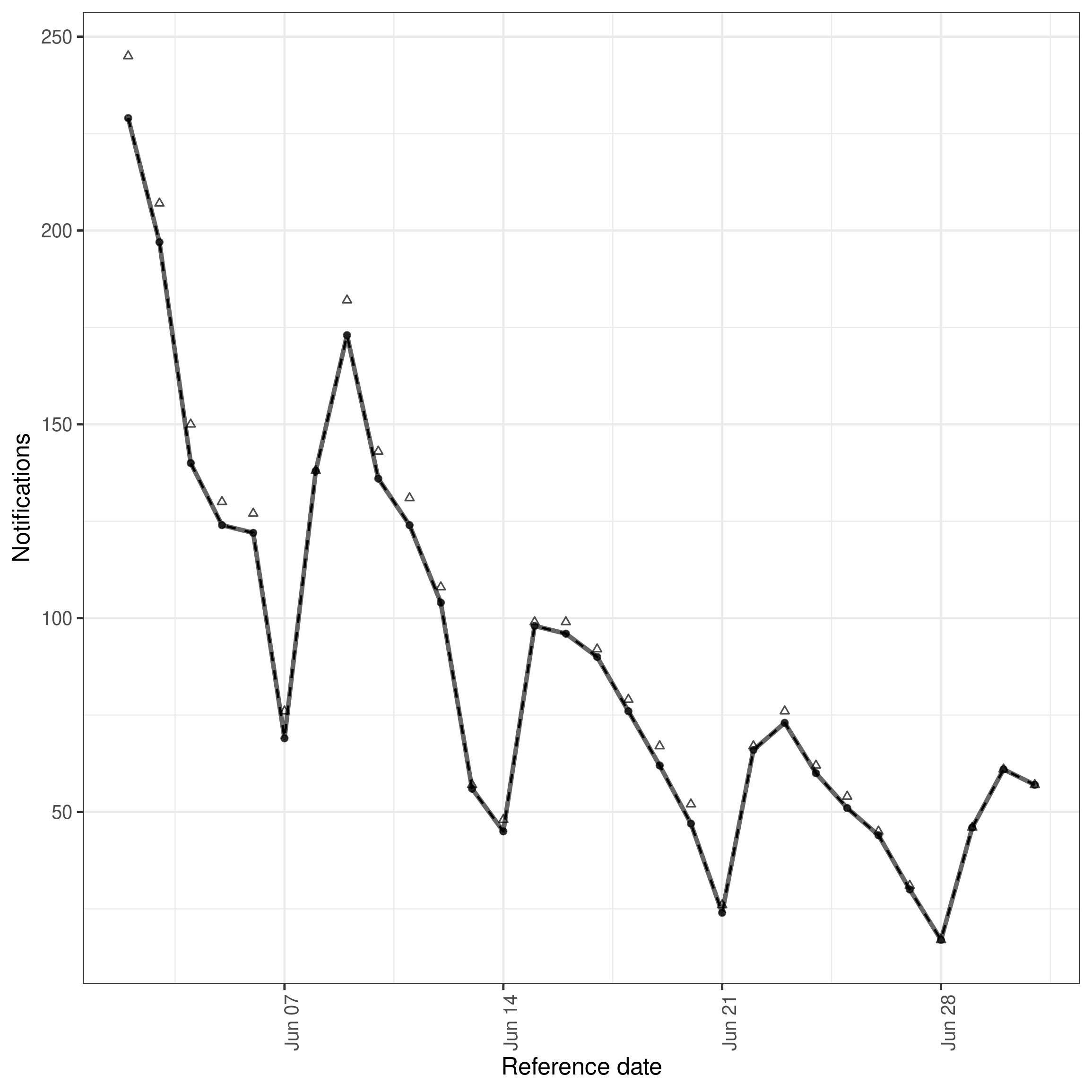
Posterior predictions for cases by date of positive test and report
To better understand the fit of the model to data we can instead plot the posterior predictions for the observed data. This is effectively the same as the plot above but instead of plotting the posterior predictions for the unobserved data we plot the posterior predictions for the observed data. This can be used to get a feel for how well the model is able to predict the observed data. Rather than using the build in plot function (by changing type = "posterior_prediction") we instead use enw_plot_pp_quantiles() so we can control the number of references dates we plot (as otherwise the plot will be quite overwhelming!).
Code
plot_select_pp_dates <- function(nowcast, dates) {
nowcast |>
summary(type = "posterior_prediction") |>
(\(x) x[reference_date %in% dates])() |>
enw_plot_pp_quantiles() +
facet_wrap(vars(reference_date), scales = "free")
}
plot_select_pp_dates(
retro_germany_nowcast,
as.Date(c("2021-05-01", "2021-05-14", "2021-06-01", "2021-07-01"))
)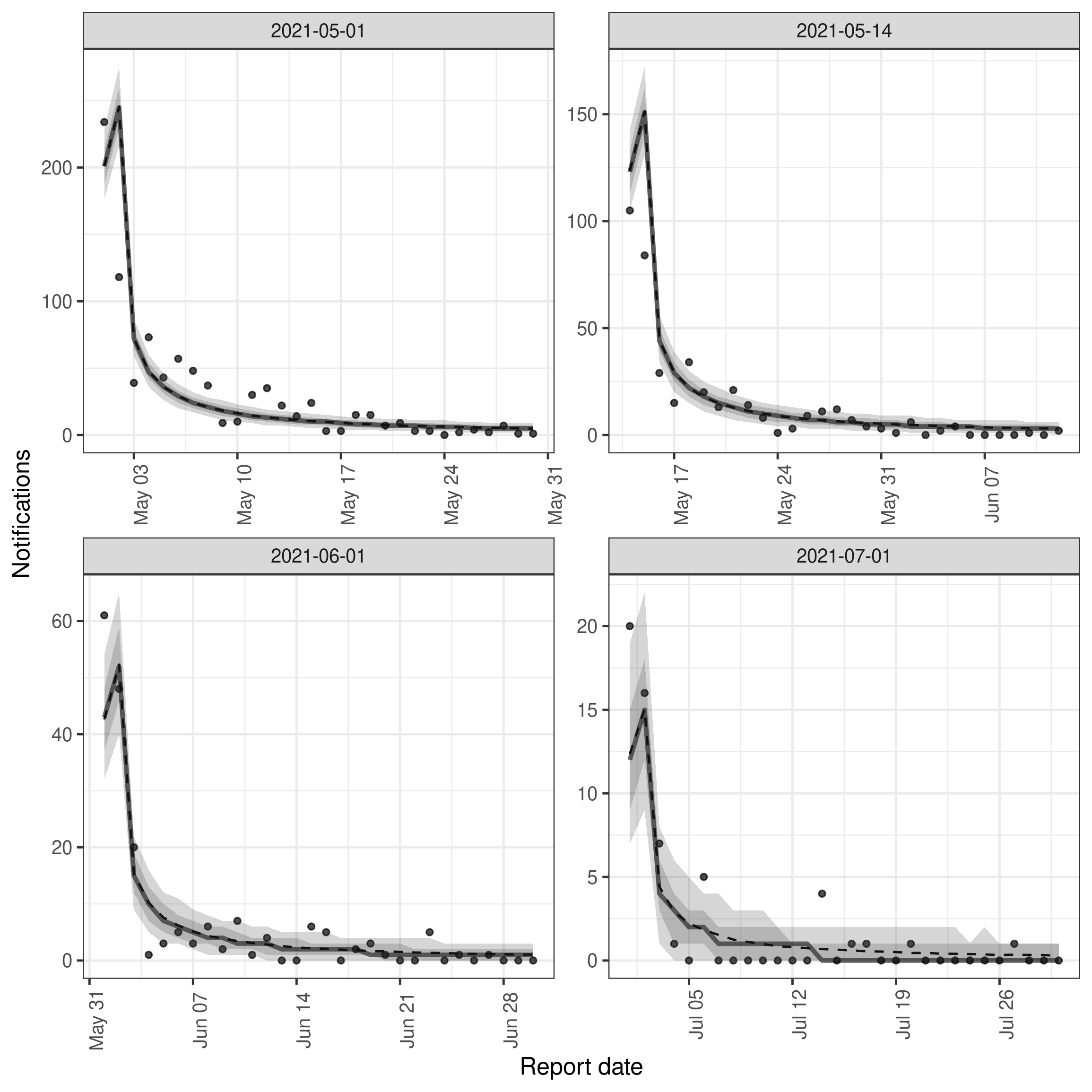
It is clear from this plot that for these example dates the model is in the main able to predict the observed data well. However, there are also some aspects of the data that the model is less well able to predict, in particular the apparent periodicity in the reporting delay. This is likely due to the model not being able to account for the day of the week reporting periodicity. This could be fixed by adding a day of the week random effect to the reporting delay model which we demonstrate in the package README.
Real-time and retrospective estimates of the effective reproduction number
A key output of the model is the posterior distribution of the effective reproduction number. We can plot this for both the real-time and retrospective data to see how the estimates change as more data becomes available. Ideally, we would hope that the real-time estimates would overlap with the retrospective estimates. This would indicate that our model is able to accurately estimate hospitalisations that will be ultimately reported based on those that have already been reported.
Code
get_rt_posterior <- function(nowcast, expectation = expectation_module) {
rt <- enw_posterior(nowcast$fit[[1]], variables = "r")
cols <- c("mean", "median", "q5", "q20", "q80", "q95")
rt[, (cols) := lapply(.SD, exp), .SDcols = cols]
rt <- cbind(
expectation(data = nowcast)$data_raw$r[, .(date)], rt
)
return(rt)
}
rt <- rbind(
get_rt_posterior(germany_nowcast)[, Data := "Real-time"],
get_rt_posterior(retro_germany_nowcast)[, Data := "Retrospective"],
get_rt_posterior(retro_nodelay_nowcast)[, Data := "Retrospective (no delay)"]
)
ggplot(rt) +
aes(x = date, col = Data, fill = Data) +
geom_line(aes(y = median), linewidth = 1, alpha = 0.6) +
geom_line(aes(y = mean), linetype = 2) +
geom_ribbon(aes(ymin = q5, ymax = q95), alpha = 0.2, linewidth = 0.2) +
geom_ribbon(aes(ymin = q20, ymax = q80, col = NULL), alpha = 0.2) +
geom_hline(yintercept = 1, linetype = 2) +
scale_y_continuous(trans = "log") +
scale_fill_brewer(palette = "Dark2", aesthetics = c("color", "fill")) +
theme_bw() +
labs(
x = "Date of infection",
y = "Effective reproduction number"
) +
theme(legend.position = "bottom")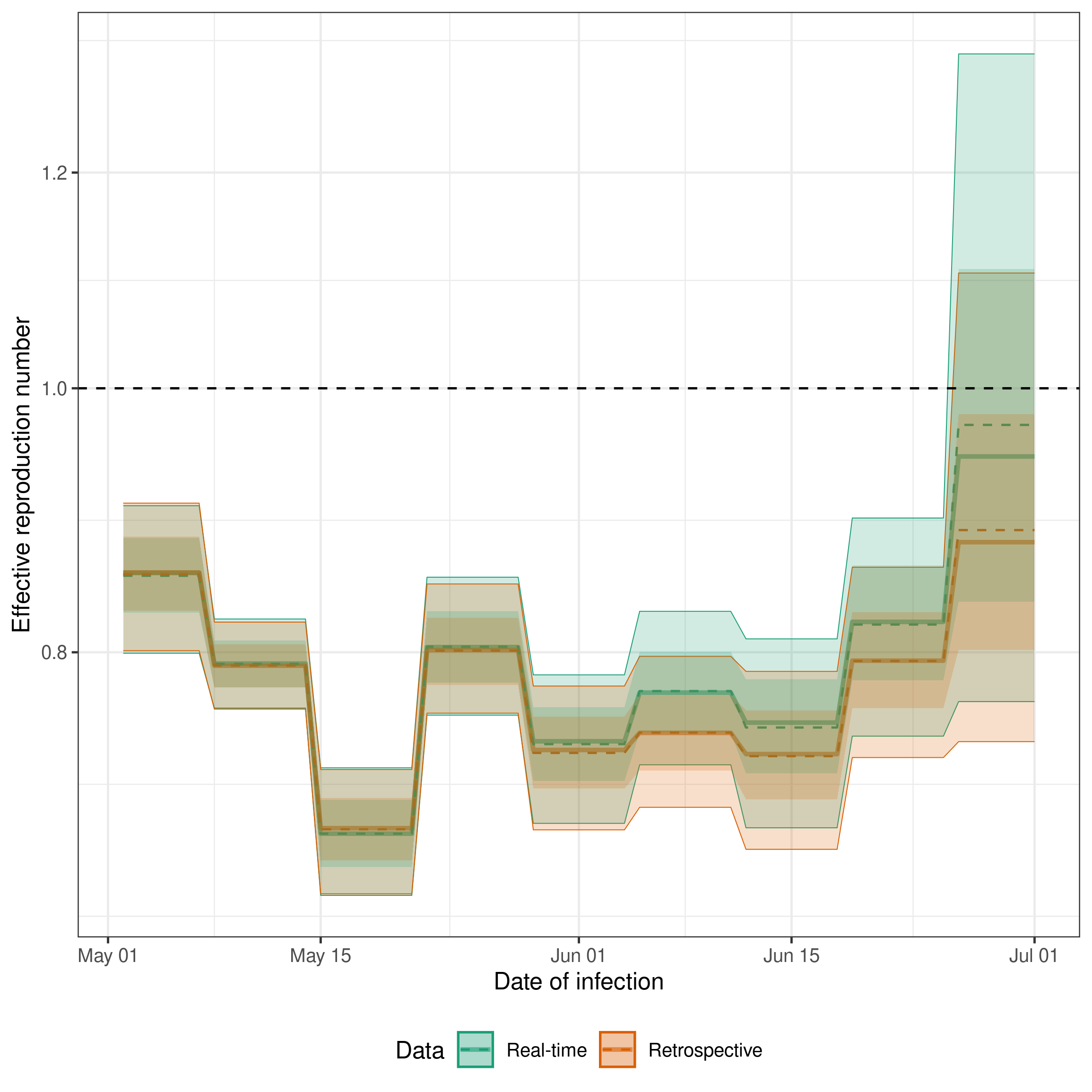
Here we see good agreement between the real-time and retrospective estimates of the effective reproduction number for dates earlier in time as we would expect given that this data should be near complete in both models. Closer to the date of estimation we see that the real-time model does a good job estimating the trend shown by the retrospective model but has a tendency to overpredict. This could be caused by changes in the reporting delay over time, by the model not being able to account for the day of the week reporting periodicity, or by the model not being able to account for the change in the underlying process over time.
The retrospective model without delay estimation (“Retrospective (no delay)”) provides a useful baseline. It uses the same expectation model but treats the latest available counts as final, skipping delay estimation entirely. This is faster to fit and can be useful when data is already complete or when the primary interest is Rt estimation rather than nowcasting. The agreement between this and the full retrospective model indicates that the delay model is not substantially biasing the Rt estimates in this example.
Estimates of the delay from testing positive to hospitalisation both in real-time and retrospectively
We can also plot the posterior distribution of the delay from testing positive to hospitalisation. This is the delay that is used to estimate the number of hospitalisations that will ultimately be reported based on the number of positive tests that have been reported. We can plot this for both the real-time and retrospective data to see how the estimates change as more data becomes available. Ideally, we would hope that the real-time estimates would overlap with the retrospective estimates. This would indicate that our model is able to accurately estimate hospitalisations that will be ultimately reported based on those that have already been reported. Note that the retrospective model without delay estimation is not included here as it does not estimate a reporting delay.
Code
extract_epinowcast_cdf <- function(nowcast) {
draws <- nowcast |>
(\(x)
x$fit[[1]]$draws(variables = c("refp_mean", "refp_sd"), format = "df")
)() |>
as.data.table()
draws[
,
cdf := purrr::map2(
`refp_mean[1]`, `refp_sd[1]`,
~ data.table(
delay = 1:30, cdf = plnorm(1:30, .x, .y) / plnorm(30, .x, .y)
)
)
]
draws <- draws[, rbindlist(cdf)]
draws <- draws[,
.(
mean = mean(cdf),
lower_90 = quantile(cdf, probs = 0.05),
upper_90 = quantile(cdf, probs = 0.95)
),
by = "delay"
]
}
nowcast_cdf <- list(
"Real-time" = germany_nowcast,
"Retrospective" = retro_germany_nowcast
) |>
map(extract_epinowcast_cdf) |>
rbindlist(idcol = "Data")
ggplot(nowcast_cdf) +
aes(x = delay, y = mean, col = Data, fill = Data) +
geom_line(size = 1.1, alpha = 0.7) +
geom_ribbon(
aes(ymin = lower_90, ymax = upper_90),
alpha = 0.25
) +
theme_bw() +
theme(legend.position = "bottom") +
scale_fill_brewer(palette = "Dark2", aesthetics = c("color", "fill")) +
guides(
fill = guide_legend(nrow = 1),
col = guide_legend(nrow = 1)
) +
labs(
x = "Delay between positive test and hospitalisation",
y = "Cumulative density function of the reporting distribution"
)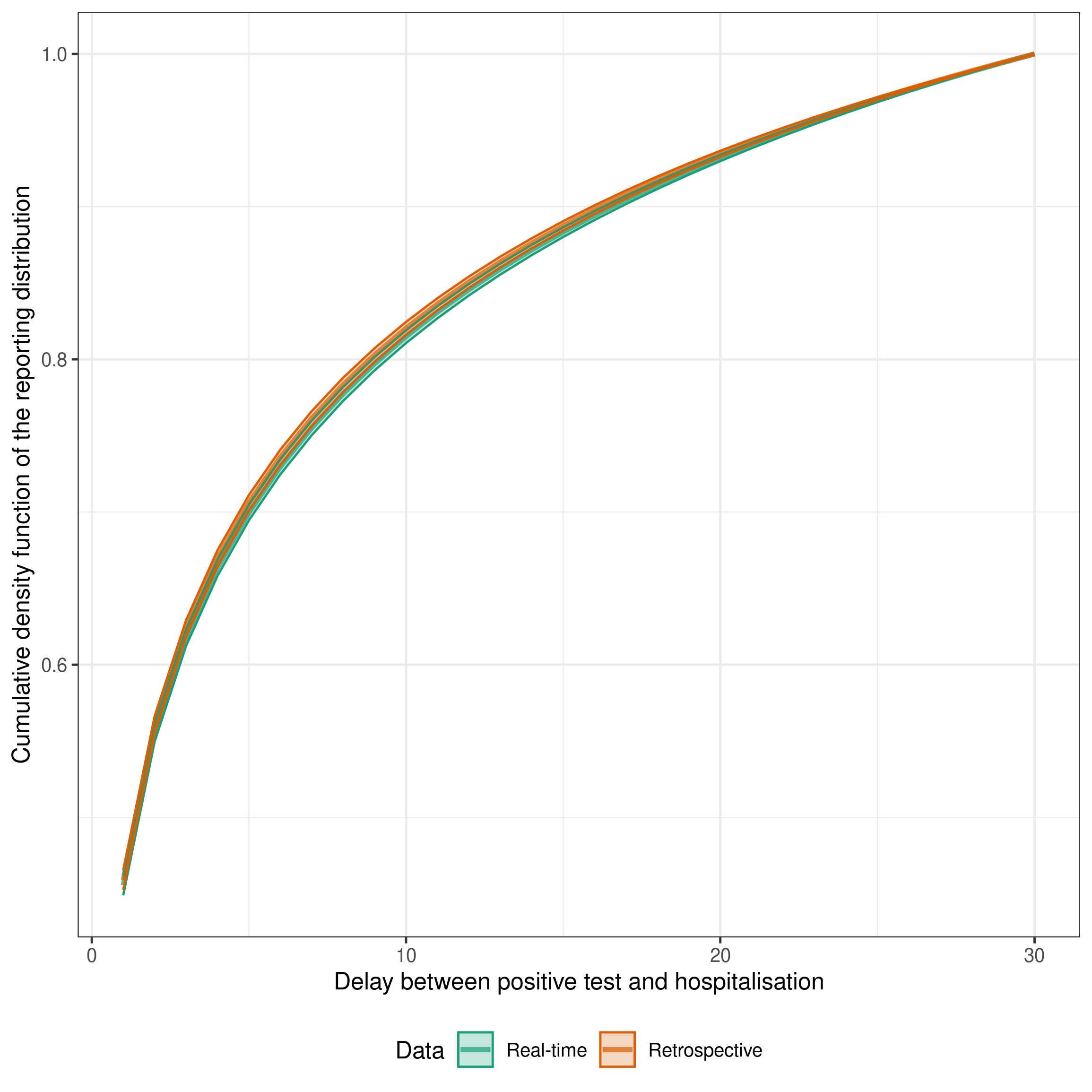
The estimates are a fairly good match indicating that the delay is being well estimated by the real-time model compared to the retrospective model and that the delay is unlikely to be changing rapidly over time in this example (as otherwise we would expect the estimated delays to diverge over time).
Estimates of the number of expected hospitalisations both in real-time and retrospectively
We can also plot the posterior predictions for the number of expected hospitalisations. This is the number of hospitalisations that would be reported if there was no observation error. We can compare this to the number of hospitalisations that are ultimately reported to see how well the model is doing.
Retrospectively, we would expect the model to fit very well to this data with any differences being due to the observation error. If this is not the case it indicates we may need to revisit our expected hospitalisation model specification to make it closer to the generative process of the data we have.
In real-time, we would expect the model to fit less well to this data as it is trying to estimate the number of hospitalisations that will ultimately be reported based on the data that is currently available (i.e. it also has to account for the reporting delay which is now partially unobserved).
Code
get_expected_infections <- function(nowcast, expectation = expectation_module) {
exp_cases <- enw_posterior(
nowcast$fit[[1]],
variables = "exp_lobs"
)
cols <- c("mean", "median", "q5", "q20", "q80", "q95")
exp_cases[, (cols) := lapply(.SD, exp), .SDcols = cols]
exp_cases <- cbind(
expectation(data = nowcast)$data_raw$observation,
exp_cases
)
return(exp_cases)
}
exp_cases <- rbind(
get_expected_infections(germany_nowcast)[, Data := "Real-time"],
get_expected_infections(retro_germany_nowcast)[, Data := "Retrospective"],
get_expected_infections(
retro_nodelay_nowcast
)[, Data := "Retrospective (no delay)"],
fill = TRUE
)
exp_cases <- latest_germany_hosp[, date := reference_date][
exp_cases,
on = "date"
]
ggplot(exp_cases) +
aes(x = date, fill = Data, col = Data) +
geom_point(aes(y = confirm), col = "Black") +
geom_line(aes(y = median), linewidth = 1, alpha = 0.6) +
geom_line(aes(y = mean), linetype = 2) +
geom_ribbon(aes(ymin = q5, ymax = q95), alpha = 0.2, linewidth = 0.2) +
geom_ribbon(aes(ymin = q20, ymax = q80, col = NULL), alpha = 0.2) +
theme_bw() +
labs(
x = "Date of positive test",
y = "Expected hospitalisations"
) +
scale_fill_brewer(palette = "Dark2", aesthetics = c("color", "fill")) +
theme(legend.position = "bottom")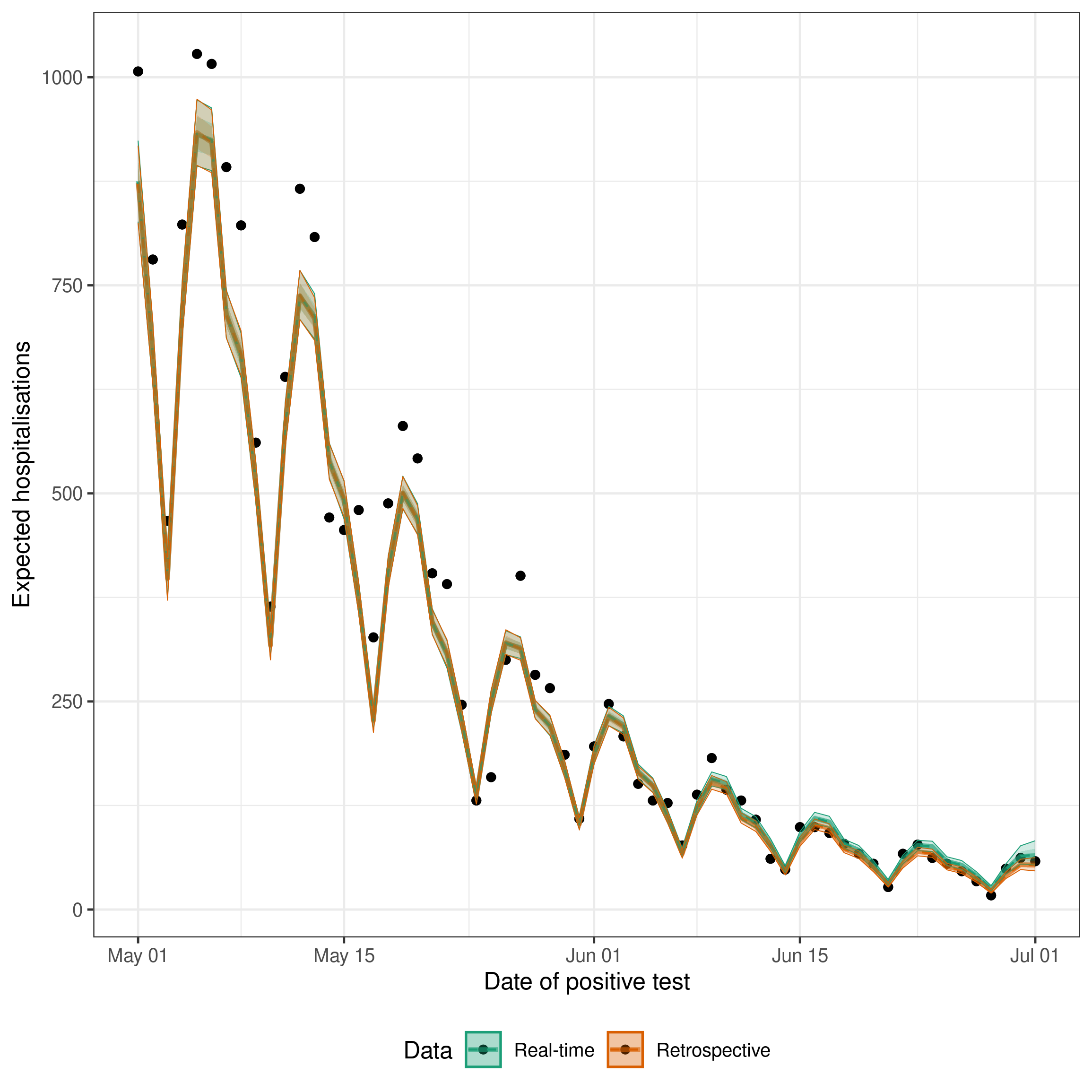
The black points here are the counts at the maximum modelled delay (latest_germany_hosp, the rolling “obs at max delay” set) — the same data the retrospective models see, which is what enw_retrospective() also uses internally.
The ribbons show posterior uncertainty on the expected observations (exp_lobs), not posterior predictive draws, so they do not include Poisson observation noise and individual points will scatter around the fitted mean even for a well-specified model.
As expected the retrospective estimates fit very well to the data. The real-time estimates are slightly less good, but still capture the recent change in trend in the data which would not be possible without a nowcast (as estimates would be biased towards underpredicting due to the delay in reporting leading to right truncation).
Wrapping up
Summary
In this case study we have shown how to use epinowcast to estimate the effective reproduction number and the expected number of latent and reported cases from right truncated data.
We have also shown how to use the package to perform retrospective and real-time nowcasts which can be a useful way of understanding the real-time performance of the model.
In addition, we demonstrated retrospective Rt estimation without delay modelling using enw_retrospective(), which provides a faster alternative when data is already complete.
Strengths
- We have used a single model to estimate the effective reproduction number and the expected number of latent and reported cases from right truncated data. This means that we have propagated uncertainty from the estimation of the effective reproduction number into the estimation of the expected number of latent and reported cases which would be difficult using a multi-stage approach.
- We have used a flexible model for the effective reproduction number which allows us to estimate the effective reproduction number over time and to incorporate uncertainty in the estimation of the effective reproduction number into the estimation of the expected number of latent and reported cases.
- We have accounted for the delay from infection to hospitalisation and the delay from hospitalisation to reporting in the estimation of the expected number of reported cases. This means that reproduction number estimates are currently indexed to the date of infection.
Limitations
- Assumed a known and static generation time distribution and latent reporting delay distribution. In reality these would need to be estimated from data, have uncertainty, and be liable to change over time.
- Used a fixed parametric reporting delay distribution. In reality this would likely vary over time and depend on things like day of the week reporting effects. This could be addressed by using a more flexible reporting delay distribution which is supported by
epinowcast. - Used an observation model that does not account for overdispersion in the number of reported cases. This could be addressed by using a more flexible observation model which is supported by
epinowcast.
Alternative packages
-
EpiNow2: A precursor to
epinowcastdeveloped by members of theepinowcastcommunity. It is a flexible toolset for real-time analysis of infectious diseases. It is less complex thanepinowcastwith a focus on robust default settings but is also less flexible. Over time its functionality will be incorporated intoepinowcast. -
epidemia: This is another flexible package for estimating the effective reproduction number and forecasting. It is designed to be more flexible than
EpiNow2, in a similar way toepinowcast, but is potentially more difficult to use. It also generally has less functionality for dealing with delays thanepinowcast. However, as it is an extension ofrstanarmit comes with a number of useful features and a familiar interface for users ofrstanarm - EpiEstim: This is a more mature package for estimating the effective reproduction number. It exploits a mathematically relationship to fit the renewal equation with uncertainty very quickly but is not currently able to handle reporting delays or other aspects of real-world data that we have discussed in this case study. It is also not able to perform nowcasts. However, it is a useful package for estimating the effective reproduction number in settings where these aspects are not important or where they can be handled by other means.